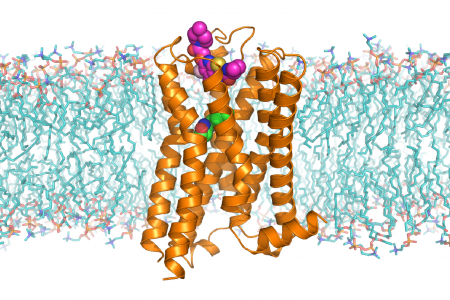
G protein-coupled receptors (GPCRs) and other transmembrane signaling proteins mediate the flow of information across lipid membranes. They enable cells to sense and respond to their environment, and they allow coordination of physiological processes across multiple organ systems in the body. We use a combination of structural biology methods, pharmacological and signaling assays, and protein engineering to investigate signaling by GPCRs, tetraspanin proteins, and other integral membrane receptors. In particular, we are investigating the antiotensin II receptor and the RXFP1 relaxin receptor to understand the molecular basis for activation and inhibition by natural and synthetic ligands.
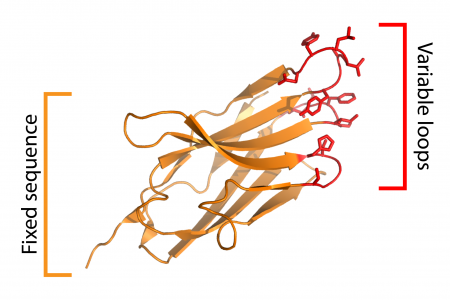
Antibody fragments and other protein affinity reagents are powerful tools for investigating biology. We are developing new methods to make antibody fragment discovery faster, simpler, and more reliable. We focus primarily on using synthetic camelid heavy chain only antibodies (“nanobodies”) as tools to study transmembrane signaling. We previously developed a nanobody library which is available for non-profit research from Kerafast: https://www.kerafast.com/item/1770/yeast-display-nanobody-library-nblib. For-profit organizations can license the library from the Harvard Office of Technology Development.
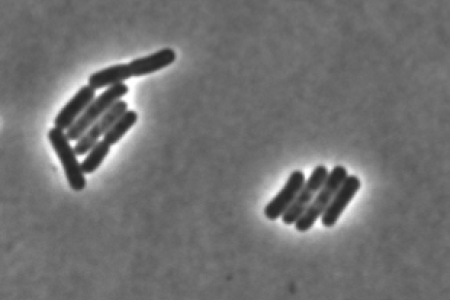
We are investigating how bacterial build and maintain their protective outer layers: the peptidoglycan cell wall, and in Gram-negative bacteria, the outer membrane. These elaborate structures are essential for bacterial growth, survival, and pathogenesis, making the cell envelope a compelling target for novel antibiotic development. We are using structural and biophysical methods to investigate how integral membrane enzymes assemble the cell wall and outer membrane, and how this process is regulated in molecular detail.